High-entropy materials (HEMs)
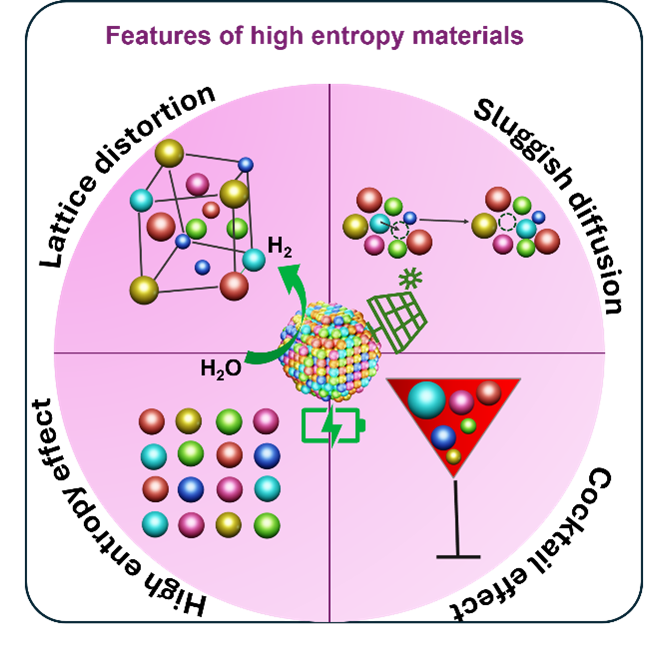
High-entropy materials (HEMs) consist of five or more principal elements randomly distributed over crystallographic lattice sites, leading to a large configurational entropy that can stabilise single-phase solid solutions. This has resulted in unique features of HEMs, including lattice distortion, sluggish diffusion, the high entropy effect, and cocktail effects. These unique features have led to significant attention to these materials in applications such as catalysis, batteries, photovoltaics, and thermoelectrics.
Among the different classes of HEMs, high-entropy chalcogenides (HEChs) remain relatively unexplored. Their synthesis is challenging because multiple metal cations often exhibit varying reactivities toward chalcogen precursors, leading to competing phases and side products. In addition, most traditional syntheses of HEMs have employed high temperatures, yielding bulk structures, while synthesis in the nano regime has been scarcely explored.
To address the above challenges, our group explores colloidal synthetic approaches for HEChs encompassing sulphides, selenides, and tellurides. Colloidal methods offer precise control over reaction parameters, while avoiding the extremely high temperatures typically required for entropy stabilisation in bulk materials. In contrast to direct one-pot synthesis, strategies such as cation exchange and seed-mediated approaches are being explored to prevent the formation of competing byproducts. In this direction, our group has developed synthetic strategies for high-entropy Cu-Zn-Sb-Ag-Bi-S NCs, which have shown superior performance as electrocatalysts for the hydrogen evolution reaction